kottke.org posts about CRISPR
MIT Technology Review’s Antonio Regalado reports on an improved gene editing technique that can rewrite DNA without actually cutting the DNA (which can damage and introduce errors into the genome). It’s called “prime editing”.
Today, in the latest — and possibly most important — of recent improvements to CRISPR technology, Liu is introducing “prime editing,” a molecular gadget he says can rewrite any type of genetic error without actually severing the DNA strand, as CRISPR does.
The new technology uses an engineered protein that, according to a report by Liu and 10 others today in the journal Nature, can transform any single DNA letter into any other, as well as add or delete longer stretches. In fact, Liu claims it’s capable of repairing nearly any of the 75,000 known mutations that cause inherited disease in humans.
From the abstract of the report:
Prime editing substantially expands the scope and capabilities of genome editing, and in principle could correct about 89% of known pathogenic human genetic variants.
See more coverage of this story at Nature, Scientific American, and Wired.
At the Salk Institute for Biological Studies, Dr. Joanne Chory is working on a project to create plants capable of storing more carbon for a longer period of time than normal plants in order to help mitigate the effects of climate change.
Suberin — also known as cork — is a naturally occurring carbon-rich substance found in plant roots. It absorbs carbon yet resists decomposition (which releases carbon back into the atmosphere), enriches soil and helps plants resist stress.
By understanding and improving just a few genetic pathways in plants, Salk’s plant biologists believe they can help plants grow bigger, more robust root systems that absorb larger amounts of carbon, burying it in the ground in the form of suberin.
The Salk team will use cutting-edge genetic and genomic techniques to develop these Salk Ideal Plants.
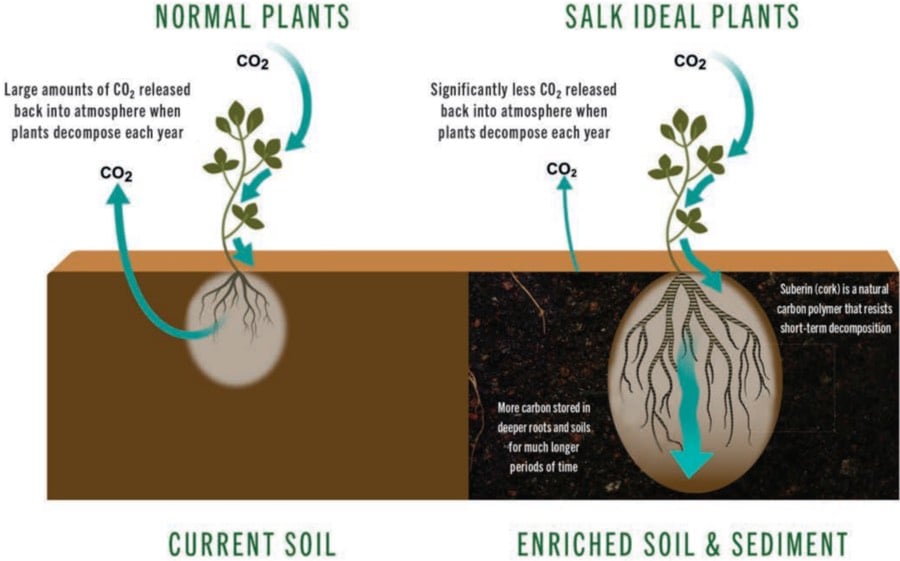
According to this piece in the Guardian on the project, one of the techniques they’re using is CRISPR, basically a genetic copy/paste system. Once the team demonstrates they can grow these larger root systems in model plants, they’ll genetically transfer that capability to the world’s largest food crops like rice, wheat, and corn.
As a bonus, the team believes that Ideal Plants will have other positive effects:
In addition to mitigating climate change, the enhanced root systems will help protect plants from stresses caused by climate changes and the additional carbon in the soil will make the soil richer, promoting better crop yields and more food for a growing global population.
This project is firmly on the wizard end of the wizards vs prophets spectrum.
In an article for MIT Technology Review, Antonio Regalado reports that researchers in Shenzhen, led by He Jiankui, are recruiting couples for a study to produce the first genetically edited human babies. They are hoping to use the CRISPR gene editing technique to “eliminate a gene called CCR5 in hopes of rendering the offspring resistant to HIV, smallpox, and cholera”.
The birth of the first genetically tailored humans would be a stunning medical achievement, for both He and China. But it will prove controversial, too. Where some see a new form of medicine that eliminates genetic disease, others see a slippery slope to enhancements, designer babies, and a new form of eugenics.
“In this ever more competitive global pursuit of applications for gene editing, we hope to be a stand-out,” He and his team wrote in an ethics statement they submitted last year. They predicted their innovation “will surpass” the invention of in vitro fertilization, whose developer was awarded a Nobel Prize in 2010.
A follow-up by the Associated Press indicates that He’s program is already underway and has resulted in a successful birth of twins earlier this month.
The researcher, He Jiankui of Shenzhen, said he altered embryos for seven couples during fertility treatments, with one pregnancy resulting thus far. He said his goal was not to cure or prevent an inherited disease, but to try to bestow a trait that few people naturally have - an ability to resist possible future infection with HIV, the AIDS virus.
He said the parents involved declined to be identified or interviewed, and he would not say where they live or where the work was done.
Update: He is now under investigation for legal and ethical misconduct.
On Sunday, the Shenzhen City Medical Ethics Expert Board said it would begin an investigation of He’s research and released a statement saying that HarMoniCare “according to our findings … never conducted the appropriate reporting according to requirements.” The former medical director of the private hospital, Jiang Su-Qi, told Southern Capital News he had no recollection of approving He’s research while he was on its ethics committee.
“These two children are the guinea pigs. They will go through their whole maturing process having not understood the risks ahead of time,” said Liu Ying of Peking University’s Institute of Molecular Medicine.
Researchers from Stanford have published a study showing that immunity might hinder the use of the CRISPR gene editing technique in humans. The Cas9 bacterial protein commonly used in CRISPR is found in and around human bodies, so many of those bodies have already built up an immunity to it. That means if you send Cas9 into a body to do some gene editing, that body’s immune system might attack and destroy it before it can do its work. Sarah Zhang wrote about the study for The Atlantic.
Porteus and his colleagues focused on two versions of Cas9, the bacterial protein mostly commonly used in CRISPR gene editing. One comes from Staphylococcus aureus, which often harmlessly lives on skin but can sometimes causes staph infections, and another from Streptococcus pyogenes, which causes strep throat but can also become “flesh-eating bacteria” when it spreads to other parts of the body. So yeah, you want your immune system to be on guard against these bacteria.
It sounds like this was something geneticists were well aware of but wasn’t common knowledge among non-technical CRISPR enthusiasts. As Chang notes, scientists are already employing strategies to route around the potential immunity roadblock:
Modify Cas9 or use a different CRISPR protein altogether: It may be possible to redesign Cas9 to hide it from the immune system or to find other bacterial proteins that can do the job of Cas9 without provoking the immune response. Many different bacteria have CRISPR systems. “We already have lots of Cas enzymes and could get many more,” George Church, a geneticist at Harvard and a founding scientific advisor of Editas, wrote in an email.
For the second part of an ongoing series, Wired asked biologist Neville Sanjana to explain CRISPR to five people with different levels of knowledge: a 7-year-old, a high school student, a college student, a grad student, and an expert on CRISPR. As I began to watch, I thought he’d gone off the rails right away with the little kid, but as soon as they connected on a personal issue (allergies), you can see the bridge of understanding being constructed.
The first installment in the series featured a neuroscientist explaining connectomes to five people.
Thousands of people die every day from malaria, a disease that is transmitted to humans solely through mosquitoes. With CRISPR, scientists can easily genetically engineer mosquitoes incapable of transmitting malaria and using a technique called gene drive, they can force that genetic change into the native mosquito population. So, should we do it?
Perfect eyesight. Curing cancer. Designer babies. Super-soldiers. Because of CRISPR, genetic engineering might make tinkering with life as easy as playing with Lego.
Imagine you were alive back in the 1980’s, and were told that computers would soon take over everything — from shopping, to dating, and the stock market, that billions of people would be connected via a kind of web, that you would own a handheld device orders of magnitudes more powerful than supercomputers.
It would seem absurd, but then all of it happened. Science fiction became our reality and we don’t even think about it. We’re at a similar point today with genetic engineering. So let’s talk about it.
Relatedly, I’m finishing up Neal Stephenson’s Seveneves right now and while it starts out as space science fiction, much of the book is concerned with the sort of genetic engineering issues discussed in the video.
Michael Specter has a truly fascinating piece in the New Yorker about CRISPR, a relatively new genetic tool for editing genes that geneticists are very excited about.
With CRISPR, scientists can change, delete, and replace genes in any animal, including us. Working mostly with mice, researchers have already deployed the tool to correct the genetic errors responsible for sickle-cell anemia, muscular dystrophy, and the fundamental defect associated with cystic fibrosis. One group has replaced a mutation that causes cataracts; another has destroyed receptors that H.I.V. uses to infiltrate our immune system.
The story has everything: the cheap copy/paste of DNA, easily editable mice, pig Hitler, “destroyer of worlds” overtones, and an incredible tale of science that could actually revolutionize (or ruin, depending on who you talk to) the world. I was shocked at how easy it is to do genetic research nowadays.
Ordering the genetic parts required to tailor DNA isn’t as easy as buying a pair of shoes from Zappos, but it seems to be headed in that direction. Yan turned on the computer at his lab station and navigated to an order form for a company called Integrated DNA Technologies, which synthesizes biological parts. “It takes orders online, so if I want a particular sequence I can have it here in a day or two,” he said. That is not unusual. Researchers can now order online almost any biological component, including DNA, RNA, and the chemicals necessary to use them. One can buy the parts required to assemble a working version of the polio virus (it’s been done) or genes that, when put together properly, can make feces smell like wintergreen. In Cambridge, I.D.T. often makes same-day deliveries. Another organization, Addgene, was established, more than a decade ago, as a nonprofit repository that houses tens of thousands of ready-made sequences, including nearly every guide used to edit genes with CRISPR. When researchers at the Broad, and at many other institutions, create a new guide, they typically donate a copy to Addgene.
And CRISPR in particular has quickened the pace. A scientist studying lung cancer mutations said of her research:
“In the past, this would have taken the field a decade, and would have required a consortium,” Platt said. “With CRISPR, it took me four months to do it by myself.”
Also recommended: Radiolab’s podcast on CRISPR from back in June.
No hunger. No pollution. No disease. Wired’s Amy Maxmen welcomes you to the age of copy and paste DNA editing and the end of life as we know it.
Genome editing started with just a few big labs putting in lots of effort, trying something 1,000 times for one or two successes. Now it’s something that someone with a BS and a couple thousand dollars’ worth of equipment can do. What was impractical is now almost everyday. That’s a big deal.
[I recently listened to Radiolab’s show on Crispr. Recommended. -jkottke]





Socials & More